What Is Battery Acid?Common Types, Specs & Safety
By NACCI Battery Technology Team·April 2026·8 min read

Battery acid — sulfuric acid (H₂SO₄) diluted with purified water — is the electrolyte that powers every lead-acid battery. Image: NACCI Battery Technology, April 2026.
Battery acid is one of the most important and least understood substances in modern energy storage. Every time your car starts, every time your solar storage system charges, every time a telecom tower stays live during a grid outage — battery acid is at work inside the cells, making it all possible. Understanding what it is, how it behaves, and what its specifications mean is essential for anyone who specifies, maintains, or sources lead-acid batteries.
In This Guide
- What is battery acid, exactly?
- How it works inside a battery
- Common types by application
- Specifications chart
- How to read specific gravity
- Safety: six critical rules
- FAQ — expert answers
What Is Battery Acid, Exactly?
Battery acid is a dilute aqueous solution of sulfuric acid (H₂SO₄) mixed with purified water. In its pure industrial form, sulfuric acid can reach 93–98% concentration. Inside a lead-acid battery, the concentration is deliberately reduced — typically to 29–40% sulfuric acid by weight — to create the electrolyte that powers the cell.
The term “battery acid” is almost always synonymous with this sulfuric acid electrolyte. It is colorless, odorless in dilute form, highly corrosive on contact with skin, and has a pH of approximately 0.8 at working concentration. It is not a simple filler — it is the active chemical participant in every charge and discharge cycle the battery completes.
“Battery acid is not passive filler. It reacts, converts, and recovers with every single charge-discharge cycle — and its concentration tells you exactly how much charge is left.”
How Battery Acid Works Inside a Cell

The sulfuric acid electrolyte sits between the positive lead dioxide plate and the negative sponge lead plate, enabling ion transfer during charge and discharge. Image: NACCI, 2026.
Inside every lead-acid battery cell, there are two types of plates: a positive plate made of lead dioxide (PbO₂) and a negative plate made of sponge lead (Pb). Both are submerged in the sulfuric acid electrolyte. The acid does not just sit there — it participates actively in the electrochemical reaction.
During discharge, the sulfuric acid reacts with both plates. The lead dioxide at the positive plate and the sponge lead at the negative plate both convert into lead sulfate (PbSO₄). In doing so, they release electrons — which is the electrical current you draw from the battery. The acid is consumed in this process and converted partly into water, which is why the electrolyte becomes less dense as the battery discharges.
During charging, the process reverses entirely. An external current drives the lead sulfate back into lead dioxide and sponge lead, and converts the water back into sulfuric acid. The electrolyte density rises. When fully charged, the acid concentration is at its peak. This reversibility is the entire reason lead-acid batteries can be recharged — and it is the battery acid that makes that chemistry possible.
Why specific gravity matters: Because the acid concentration changes with state of charge, you can use a hydrometer to measure the electrolyte density and get a direct reading of how much charge remains. A fully charged cell reads approximately 1.265–1.285 g/cm³. A discharged cell reads close to 1.100–1.150 g/cm³. This is one of the most reliable diagnostic tools available for flooded lead-acid batteries.
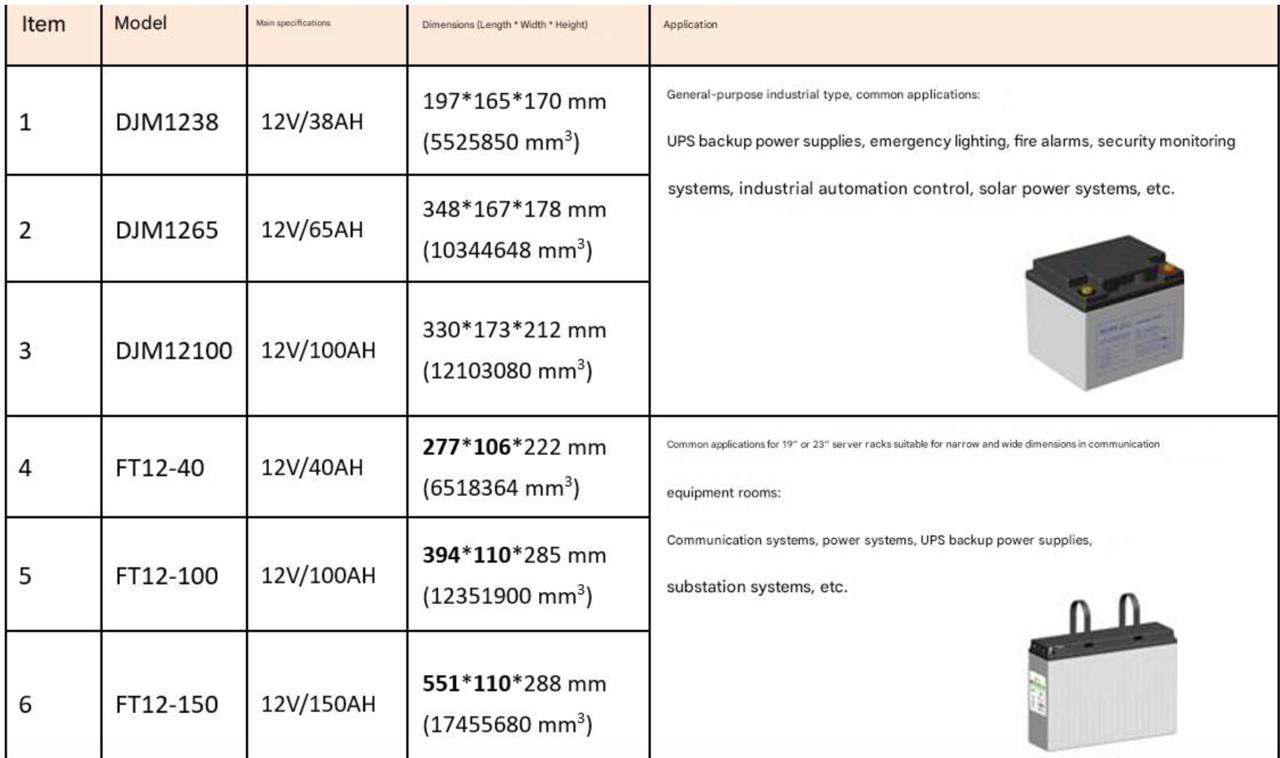
Common Types of Battery Acid by Application
Not all lead-acid batteries use the same acid concentration. The strength of the electrolyte is engineered for the battery’s specific duty cycle, plate thickness, operating temperature, and expected service life. Here are the most common types and where they are used.
Table 1 — Battery Acid Types by Application
| Battery Type | Electrolyte Type | H₂SO₄ Concentration | Specific Gravity (Fully Charged) | Common Application |
|---|---|---|---|---|
| Automotive / SLI | Flooded sulfuric acid | ~35% by weight | 1.260–1.280 | Car, truck, motorcycle starting batteries |
| Deep Cycle / Traction | Flooded sulfuric acid | ~37–38% by weight | 1.260–1.280 | Forklifts, golf carts, industrial vehicles |
| Stationary / UPS | Flooded sulfuric acid | ~30–32% by weight | 1.215–1.240 | Backup power, telecoms, data centers |
| AGM (Absorbed Glass Mat) | Immobilized H₂SO₄ in glass fiber mat | ~36–37% by weight | 1.280–1.310 | Start-stop vehicles, high-demand automotive |
| Gel Cell (VRLA) | H₂SO₄ + fumed silica gel | ~32–36% by weight | 1.240–1.280 | Wheelchairs, solar storage, telecoms |
| VRLA (Valve Regulated) | Recombinant sealed design | ~36–38% by weight | 1.300–1.320 | UPS, emergency lighting, security systems |
Battery Acid Specifications: Reading the Data
When sourcing battery acid or evaluating a battery’s electrolyte, these are the specifications you will encounter on technical datasheets. Understanding each one helps you assess quality, predict performance, and diagnose problems in the field.
A standard battery acid specification sheet covers density, purity, concentration, sulfate content, and impurity limits. These parameters directly determine electrolyte quality and battery cycle life. Image: NACCI Battery Specifications, 2026.
Table 2 — Battery Acid Key Specifications Explained
| Specification | Typical Value | Unit | What It Means in Plain English | Why It Matters |
|---|---|---|---|---|
| H₂SO₄ Concentration | 29–40% | % by weight | The fraction of sulfuric acid in the solution | Determines ion conductivity and voltage output |
| Specific Gravity (SG) | 1.200–1.320 | g/cm³ | How dense the acid is compared to pure water (1.000) | Direct indicator of state of charge; measured with a hydrometer |
| pH | ~0.8 (working solution) | pH scale | How acidic the electrolyte is — very low pH means very acidic | Confirms acid strength; relevant for material compatibility |
| Purity (Battery Grade) | ≥ 99.5% | % purity | The fraction of actual sulfuric acid, excluding impurities | Impurities corrode plates and reduce cycle life dramatically |
| Iron (Fe) Content | ≤ 10 ppm | ppm | Iron trace in the acid solution | Iron accelerates internal self-discharge — must be near zero |
| Chloride (Cl⁻) Content | ≤ 1 ppm | ppm | Chloride ion contamination | Chlorides corrode lead plates and collectors severely |
| Manganese (Mn) Content | ≤ 0.5 ppm | ppm | Manganese trace in the solution | Manganese deposits damage the positive plate coating |
| Color | Colorless / very slight yellow | Visual | Pure battery-grade acid is clear; yellow tint indicates impurities | First visible quality check before filling |
| Water Quality | Distilled / deionized | — | The water used to dilute the acid must be purified | Tap water minerals cause sulfation and early failure |
The most important spec most buyers overlook: Iron and chloride content. Even 15 ppm of iron in the electrolyte can reduce a battery’s cycle life by 20–30% through accelerated self-discharge. Battery-grade sulfuric acid is specifically refined to remove these metallic impurities. Never substitute industrial-grade acid for battery-grade — the impurity profile is completely different and will destroy the plates within months.
Specific Gravity Reference: State of Charge
Table 3 — Specific Gravity vs. State of Charge (12V Flooded Lead-Acid)
| Specific Gravity at 27°C | State of Charge | Voltage (12V Battery) | Recommended Action |
|---|---|---|---|
| 1.265–1.285 | 100% — Fully Charged | 12.60–12.80V | Ready for use |
| 1.225–1.260 | 75% — Partial Charge | 12.40–12.60V | Recharge soon |
| 1.190–1.225 | 50% — Half Charge | 12.20–12.40V | Recharge required |
| 1.155–1.190 | 25% — Low | 12.00–12.20V | Recharge immediately |
| Below 1.120 | 0% — Discharged | Below 11.80V | Risk of sulfation — charge without delay |
Temperature correction: Specific gravity readings vary with temperature. For every 1°C above the reference temperature of 27°C, add 0.0007 to your reading. For every 1°C below 27°C, subtract 0.0007. A hydrometer reading at 10°C that shows 1.250 corrects to 1.250 − (17 × 0.0007) = 1.238 at 27°C — a meaningful difference when diagnosing battery state of charge in cold climates.
Battery Acid Safety: Six Rules That Cannot Be Compromised
Sulfuric acid at battery working concentrations (29–40%) causes immediate chemical burns on contact with skin and permanent eye damage with minimal exposure. These six rules are non-negotiable when working with battery acid or batteries with liquid electrolyte.
1
Always add acid to water — never water to acid
Mixing concentrated sulfuric acid with water is highly exothermic — it releases significant heat instantly. Adding water to acid can cause violent spattering of hot acid. The rule is: acid into water, always. A useful memory aid: “Do as you ought to — add the acid to the water.”
2
Wear full PPE — every time, without exception
Acid-resistant gloves, full face shield (not just safety glasses), rubber apron, and closed-toe boots are required when handling battery acid or maintaining flooded batteries. Face and eye injuries account for approximately 50% of all lead-acid battery handling injuries.
3
Neutralize spills immediately with baking soda
Sodium bicarbonate (baking soda) neutralizes sulfuric acid quickly. Sprinkle generously over the spill area until fizzing stops completely. Absorb the neutralized material with an inert absorbent, then dispose as hazardous waste according to local regulations. Flush the area with water afterward.
4
Only use distilled or deionized water for top-up
When electrolyte levels drop in a flooded battery, only add distilled or deionized water — never tap water. Minerals in tap water (calcium, magnesium, chloride) cause internal short-circuits and accelerated sulfation. Never add acid to a battery that has simply dried out from evaporation — only add acid if electrolyte was physically spilled.
5
Work in a well-ventilated area to avoid hydrogen gas
Charging a lead-acid battery produces hydrogen gas, which is explosive. Overcharging also produces hydrogen sulfide — colorless, highly toxic, and heavier than air. Always charge and work with batteries in well-ventilated spaces. Never charge batteries in sealed enclosures without proper ventilation.
6
Flush skin and eye contact immediately with running water
In the event of acid contact with skin or eyes, flush immediately with large amounts of running water for a minimum of 15–20 minutes. Remove contaminated clothing while flushing. Seek medical attention immediately for any eye contact — do not wait to see if symptoms develop.
Frequently Asked Questions
Is the acid inside lithium batteries the same as in lead-acid batteries?
No — and this is an important distinction. Lithium-ion and LiFePO4 batteries do not use sulfuric acid. Their electrolyte is typically a lithium salt dissolved in an organic solvent (such as lithium hexafluorophosphate in ethylene carbonate/dimethyl carbonate). This is not “battery acid” in the traditional sense. The hazards are different: lithium battery electrolyte is flammable rather than caustic, and the primary risk is thermal runaway, not chemical burns. The term “battery acid” specifically refers to the sulfuric acid electrolyte in lead-acid chemistry.
Can I add tap water to a battery that is low on electrolyte?
No — only distilled or deionized water should ever be added to a battery. Tap water contains dissolved minerals including calcium, magnesium, iron, and chloride. Even in small concentrations, these impurities cause internal corrosion, sulfation, and irreversible capacity loss. Distilled water is inexpensive and widely available — there is no practical reason to substitute tap water. Also note: only add water, not acid, unless electrolyte was physically spilled from the battery.
What happens if battery acid concentration gets too high or too low?
Both extremes are damaging. Too low (over-diluted) reduces voltage capacity, lowers ion conductivity, and in cold climates can allow the electrolyte to freeze — rupturing the battery case. Too high (over-concentrated) accelerates corrosion of the lead plates, produces excessive heat during charging, and shortens cycle life significantly. The optimal working concentration (29–40% by weight, specific gravity 1.265–1.285 when fully charged) is a precisely engineered balance designed for maximum performance and longevity.
How do AGM and Gel batteries handle the acid differently from flooded batteries?
In AGM batteries, the sulfuric acid is absorbed into fine fiberglass mats between the plates. This immobilizes the electrolyte, eliminating the risk of spill, enabling sealed construction, and allowing the battery to operate in any orientation. The acid chemistry is identical — the difference is physical containment. In Gel batteries, the sulfuric acid is mixed with fumed silica to form a semi-solid gel. This further reduces acid movement and makes the battery highly resistant to deep discharge and vibration. Both designs are VRLA (Valve Regulated Lead-Acid) — they recombine internally generated gases rather than venting them, which is why they are maintenance-free.
What is sulfation, and how does battery acid relate to it?
Sulfation is the formation of large, hard lead sulfate crystals on the battery plates — the most common cause of premature lead-acid battery failure. When a battery is left in a deeply discharged state, the lead sulfate that forms during normal discharge begins to crystallize and harden. These crystals do not reverse during recharging — they permanently reduce the plate surface area available for electrochemical reactions, cutting capacity and raising internal resistance. The connection to battery acid: sulfation is worst when the electrolyte concentration is very low (i.e., deeply discharged), because there are insufficient sulfate ions in solution to maintain normal reversible reactions. Keeping batteries charged and the electrolyte at proper concentration prevents sulfation.
NACCI Battery Technology Team — Expert guides on battery chemistry, specifications, and industrial applications. All technical values referenced reflect BCI, SAE J537, and IEC standards as applicable. Published April 2026.
